| |
Did you know that around half of
all people age 70+ hear poorly, as if they have earplugs in their
ears? At the same time, unfortunately, falling is also a common
danger for a lot of older people, because dizziness and balance
problems limit their ability to move about safely. These two
problems hearing loss and balance disorders often originate
from the same source: the loss of sensory cells (known
as hair cells) from the
inner
ear.
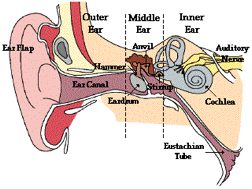
The inner ear is encased in bone
just beyond the innermost part of the
ear
canal
and is made up of two functional components known as the
cochlea
and the
vestibule
. The cochlea is a small snail-shaped
organ that allows you to hear. At birth
it contains approximately 16,000
sensory
hair cells
in four single-file rows with surrounding
rows of
supporting
cells
, which all wind around the cochleas
spiral for a little more than an inch.
Hair cells were given their name because
their function depends on a bundle
of microscopic hair-like projections
that extends up into fluid over the
cells surface.
Function
- Sound waves cause back-and-forth vibrations of the hair bundles in the cochlea
- Head movements cause similar movements of the bundles in the balance sensors of the vestibule.
Hearing
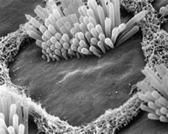 In the cochlea, each hair cell is most sensitive to a slightly different frequency of sound. A hair cell closer to the
eardrum
will be more sensitive to a higher frequency than the next cell further away, and when stimulated by sound will give rise to our perception of high pitch. The sensitivity of these hair cells and the extremely precise microscopic order of hair cell positions and nerve connections in the cochlea allow us to differentiate sound pitch so well that we can automatically distinguish between very similar words in speech, such as cat, bat, that, mat, sat, fat, and hat. We dont really think about our hearing until there is a problem. In the cochlea, each hair cell is most sensitive to a slightly different frequency of sound. A hair cell closer to the
eardrum
will be more sensitive to a higher frequency than the next cell further away, and when stimulated by sound will give rise to our perception of high pitch. The sensitivity of these hair cells and the extremely precise microscopic order of hair cell positions and nerve connections in the cochlea allow us to differentiate sound pitch so well that we can automatically distinguish between very similar words in speech, such as cat, bat, that, mat, sat, fat, and hat. We dont really think about our hearing until there is a problem.
And Balance
Similarly, most of us dont think about our balance unless theres a problem. Mostly, sensory hair cells in the vestibular portion of our inner ear operate in the background, seldom coming to our conscious attention, but their function is crucial for the stability of our visual world and our ability to move about without constantly falling. Heres how: Whenever our head moves, that causes the deflection of hair bundles in the vestibule. Those cells, in turn, send nerve signals into the brain to control an extremely fast reflex that automatically and unconsciously moves our eyes to compensate for the exact head movement that occurred, so our visual world appears to remain stable. If we did not have that reflex control, each time our head moved our visual images of the world would appear to slide around like video images do when a camera is moved too quickly. Other hair cells in the vestibule continually sense the pull of gravity and control other unconscious reflexes that allow us to stand and stay upright even when we move very quickly.
When It's Gone, It's Gone
Sadly, when sufficient numbers of hair cells are lost or damaged, the result
is permanent hearing loss known as nerve deafness or permanent balance impairments.
Old age is a common way for these conditions to arise, but hair cells can also
be damaged or killed by loud sounds, infections, head injuries, and some drugs.
Humans appear to lose the ability to produce more hair cells after they are
born, consequently hair cell damage is usually irreversible in humans.
This is not the case, however, across the animal kingdom. Sharks, fish, amphibians,
reptiles, and birds all have the ability to make new hair cells throughout
life and they can regenerate lost hair cells that are damaged. That occurs
within a few days after those animals have experienced the same kinds of deafness
and imbalance that would be permanent for us. Their regeneration mechanisms
allow them to quickly regain hearing and balance sensitivity.
If there were some way to stimulate hair cell regrowth in human ears it might be possible
to restore hearing and balance to the millions of people suffering from hearing
loss, dizziness, and other problems today. Dr. Jeffrey Corwin of the University
of Virginia School of Medicine is investigating mechanisms of hair cell regeneration
that could do just that.
Sharks
 Growing up near the woods and the ocean, Dr. Corwin always had a love for animals and a particularly strong interest in marine creatures. This passion and led him to study biology as an undergraduate college student and eventually brought him to the Marshall Islands for doctoral research on sharks. At the time, sharks were assumed to have primitive ears since they are an ancient fish, whose close relatives have been around for hundreds of millions of years. The vestibular parts of the shark ear are similar to the human ear, but the shark ear lacks the cochlea that we use for understanding speech. While working at a laboratory on Enewetak Atoll in the Marshall Islands in his first year as a research student, Dr. Corwin had access to a shark infested lagoon and lots of time to pursue his own research interests. He became curious about whether young sharks had the same number of sensory cells as older sharks. To try to figure this out, he caught
blacktip reef sharks
and preserved their inner ears. He brought the inner ears back to the laboratory and found that young sharks had about 20,000 sensory cells in one part of the inner ear compared to the 240,000 found in that same part older sharks. Growing up near the woods and the ocean, Dr. Corwin always had a love for animals and a particularly strong interest in marine creatures. This passion and led him to study biology as an undergraduate college student and eventually brought him to the Marshall Islands for doctoral research on sharks. At the time, sharks were assumed to have primitive ears since they are an ancient fish, whose close relatives have been around for hundreds of millions of years. The vestibular parts of the shark ear are similar to the human ear, but the shark ear lacks the cochlea that we use for understanding speech. While working at a laboratory on Enewetak Atoll in the Marshall Islands in his first year as a research student, Dr. Corwin had access to a shark infested lagoon and lots of time to pursue his own research interests. He became curious about whether young sharks had the same number of sensory cells as older sharks. To try to figure this out, he caught
blacktip reef sharks
and preserved their inner ears. He brought the inner ears back to the laboratory and found that young sharks had about 20,000 sensory cells in one part of the inner ear compared to the 240,000 found in that same part older sharks.
 This was a surprising discovery. First, there were many more sensory cells in the primitive inner ears of older sharks than had ever been described for any other species. Second, there was almost no previous evidence to indicate that sensory cells could be added to an ear after birth. Nerve deafness was assumed to be permanent because scientists thought that hearing and balance sensory cells could not be produced after birth. Since this new evidence showed that sharks could produce hundreds of thousands of sensory hair cells during their lives, it could mean that mammals could have that capability as well. Even if they didnt have such an innate capability, exploring the biological processes that gave rise to new hair cells in sharks might provide a great opportunity for scientists to learn strategies to stimulate hair cell production and develop future treatments for hearing loss and balance problems in humans. This was a surprising discovery. First, there were many more sensory cells in the primitive inner ears of older sharks than had ever been described for any other species. Second, there was almost no previous evidence to indicate that sensory cells could be added to an ear after birth. Nerve deafness was assumed to be permanent because scientists thought that hearing and balance sensory cells could not be produced after birth. Since this new evidence showed that sharks could produce hundreds of thousands of sensory hair cells during their lives, it could mean that mammals could have that capability as well. Even if they didnt have such an innate capability, exploring the biological processes that gave rise to new hair cells in sharks might provide a great opportunity for scientists to learn strategies to stimulate hair cell production and develop future treatments for hearing loss and balance problems in humans.
When it came time for Dr. Corwin to establish his own laboratory, he decided to continue research on hair cell production and regeneration. Dr. Corwin focused on the vestibular system rather than the cochlea because the vestibular system is very similar between mammalian and non-mammalian species, whereas the cochlea are quite different. Dr. Corwin started conducting experiments to see whether hair cells could be regenerated in amphibians such as salamanders and frogs and found evidence that replacement hair cells were developing from cells produced when neighboring supporting cells divided to form two new cells.
Collaboration
At about the same time his friend and frequent collaborator, Dr.
Doug Cotanche, who is now at Boston University, was studying
hearing loss in chickens. Dr. Cotanche played loud sounds to
young chickens and then preserved their ears in order to assess
how hair cell damage was occurring. He preserved the ears of
some birds immediately after exposure to the loud sound, and
he let others recover for 10 days before preserving their ears.
Remarkably, he found that ears preserved 10 days after the loud
sounds no longer appeared to have the hair cell damage that was
found in birds whose ears had been preserved immediately. This
could only mean one thing -- that birds were able to repair hair
cell damage in the cochlea. The results also suggested that birds
could make new, replacement hair cells in a similar way to the
sharks, salamanders, and frogs Dr. Corwin had been studying.
Dr. Corwin and Dr. Cotanche then collaborated to conduct experiments
to either confirm or refute their hypothesis that the reversal
of hair cell damage was due to the production of replacement
hair cells. For this they gave birds a radioactive form of
thymine
, one of the four building blocks of
DNA,
known as tritiated
thymine just after damaging the hair cells with
loud sound. If the tritiated thymidine was incorporated into the damaged
region, that would mean that cells in the birds cochlea
had replicated their DNA and divided to produce new cells, as
they repaired the damage. In fact, when Drs. Corwin and Cotanche
looked at the damaged region of the cochlea in these birds they
found tritiated thymine had been incorporated into hair cells
and supporting cells in the regions that had been repaired. This
proved that birds were capable of producing new hair cells at
a later point in life. Other scientists have replicated these
results in similar experiments. In subsequent experiments,
Dr. Corwin, Dr. Cotanche, and their colleagues confirmed that
the replacement hair cells were produced from nearby supporting
cells.
Other scientists showed that the new hair cells were able to
successfully connect to the nervous system to transmit signals,
enabling birds to recover their hearing and balance even after
experiencing severe nerve deafness.
What About Mammals?
Dr. Corwin wondered what it was about the mammalian ear that prevents the effective replacement of lost hair cells. Jason Meyers, who is now a biology professor at Colgate University; Jared Christophel, who is an ear, nose, and throat surgeon; and Joseph Burns, who is a biomedical engineer, worked with Dr. Corwin to investigate the hypothesis that a difference in the way the cells in mammalian ears were attached to their neighbors was restricting hair cell regeneration. Every cell has a
cytoskeleton
- sort of a microscopic scaffold built out of special proteins, which give cells their structure. Our skin and the linings of internal organs are made of sheets of cells that are attached tightly to all their neighboring cells. In those cellular sheets, proteins of the cytoskeleton form a belt, encircling each cell at the point where it holds onto its neighboring cells. In the ears of fish, amphibians, and birds, the cytoskeletal belts at the junction between neighboring cells remain thin throughout the life of the animal.
In mammalian ears, however, those belts thicken greatly during the first few weeks of life. Dr. Corwin and his coworkers hypothesize that this thickening prevents the type of hair cell regeneration that occurs innately in non-mammalian species.
In a set of recent experiments, Joseph Burns and Dr. Corwin have been using drug treatments, gene delivery, and other manipulations to trim the thickness of the cytoskeletal belts in the ears of mice. They want to see whether hair cell regeneration might be possible when those belts are reduced. The results from these experiments look promising so far, but need to be carefully replicated.
The Counting Problem
Another major problem faced by researchers in this field is difficult and time consuming micro-dissection that must be done to get enough hair cells for experiments. The inner ear is a very small and complicated structure and it can take hours to extract even a sample of the several thousand hair cells it contains. This differs from the usual case, in which biomedical scientists are able to grow large numbers of cell lines for experimentation. This is done by placing cells in a sterile dish that contains the nutrients needed for growth, and by keeping the dish in an incubator at body temperature. Such cultured cells grow, divide, and continually increase in number just as if they were in the body.
Dr. Corwin, his colleague, Dr. Zhengqing Hu, who is now a Professor at Wayne State University Medical School, and other scientists have sought to discover ways to produce hair cells in culture. In order to develop a method for growing hair cells, Drs. Hu and Corwin took supporting cells from the ears of chicken
embryos
and placed them in culture with nutrients the cells use to grow. Once the cells had grown to large numbers, Drs. Hu and Corwin transferred small samples of the cells in a new dish and began to continuously rock the dish back and forth to keep the cells moving around so much that they could not hold onto the surface of the dish. The cells began to hold onto each other, aggregating and forming a mass of cells. It turns out that this was all the cells needed to cause them to change to a state from which they could specialize as bona fide hair cells and supporting cells. Dr. Corwin and other scientists are continuing this hair cell culture work, because such cells would be incredibly valuable as a tool for accelerating the pace of discoveries into hair cells, hearing loss, imbalance and new treatments.
Futures
Dr. Corwins research continues along several avenues. His main focus is to understand the mechanisms of hair cell regeneration and identify exactly what limits hair cell regeneration in humans and other mammals. He hopes that someday in the future scientists will be able to develop drug therapies to overturn the limits to regeneration in the ear and elsewhere in our bodies, so that the mammalian ear revert to a state in which it can produce replacement hair cells and other tissues can be caused to produce replacement cells as well. Such regenerative treatments could reverse hearing loss, loss of balance, and other neurological and sensory deficits in the many millions of people affected by these conditions, just as the innate mechanisms in fish, amphibians and birds do automatically.
Dr. Jeffrey Corwin is a Professor of
Neuroscience
at the University of Virginia School of Medicine studying hair cells and the mechanisms of regeneration in mammals and non-mammals. He remembers always being fascinated by animals, biology, and nature videos as a kid and decided to pursue a career in neuroscience as a result. When not in the lab, Dr. Corwin likes to go sailing, fishing, bicycling, and likes to work with his hands. One of the reasons Dr. Corwin loves his job so much is that he gets to do creative, hands-on work to explore interesting questions, and work with bright and highly motivated young people, as he and they learn new things every day.
To learn more:
- Burns, J. et al. (2008) Reinforcement of Cell Junctions Correlates With the Absence of hair Cell Regeneration in Mammals and Its Occurrence in Birds. Journal of Comparative Neurology, 511: 396-414.
- Hu, Z-Q. and Corwin, J. (2007) Inner ear hair cells produced in vitro by a mesenchymal-to-epithelial transition. Proceedings of the National Academy of Sciences, 104(42): 16675-16680
- Meyers, J. et al. (2009) Hair Cell Regeneration. Encyclopedia of Neuroscience, 4: 1005-1013.
- Meyers, J. and J. Corwin. Morphological Correlates of Regeneration and Repair in the Inner Ear. in Popper, A. ed., et al. Hair Cell Regeneration, Repair, and Protection. Springer Handbook of Auditory Research #33: 2008
For more information:
Rebecca Kranz with Andrea Gwosdow, PhD at Gwosdow Associates
HOME | ABOUT | ARCHIVES | TEACHERS | LINKS | CONTACT
All content on this site is © Massachusetts
Society for Medical Research or others. Please read our copyright
statement — it is important. |
|
|
Dr. Corwin and his collaborators

Dr. Jeffrey T. Corwin

Dr. Douglas A. Cotanche

Dr. Jason Meyers

Mr. Joseph Burns

Dr. Jared Christophel

Dr. Zhengqing Hu
Sign Up for our Monthly Announcement!
 Subscribe to all of our stories! Subscribe to all of our stories!
 Follow us on Twitter! Follow us on Twitter!
What A Year! is a project of the Massachusetts
Society for Medical Research.
Related Videos

Three interesting videos of Dr. Corwin's work from the Journal of Neuroscience:
Time-lapse movie
This one links to several very good movies about hair cells and hearing that Professor A. James Hudspeth of The Rockefeller University produced:
A Video about Hair Cells
This link contains a cool video of an outer hair cell
responding to the envelope of amplitude of the song Rock Around the Clock. The
video was produced by Professor Jonathan Ashmore of University
College, London:
Hair Cell Response
|
|

