| |
The human body runs on sugar. Not just any sugar, though. There are many types of sugars, both simple and complex, in the body. In particular, the human body needs the simple sugar glucose. Glucose drives the process of cellular respiration where cells convert glucose into energy. To survive, our bodies must ingest sufficient glucose and the glucose we ingest must be available for our cells to use.
Getting In Is Not So Easy
In order to be used by cells after digestion, glucose must first be allowed entry into cells. A glucose molecule cannot simply enter the cells by itself because cells do not recognize single glucose molecules. Instead, as with many things in the body, there is a whole process. A protein called a receptor sits on the cell membrane (The membrane is the layer around the cell that acts as the cells guardian, only letting chosen things into the cell or out of it. Teachers use the image of a plastic bag with small holes. Those holes are only unlocked for certain molecules to go in or out). The particular receptor we are interested in sits on the cell membrane and binds to the protein called insulin. [If you are a regular reader of What A Year! you know about insulin, because it is important in the disease diabetes and we have reported on it before (e.g., in April of 2009 in Sugar High.)].
When insulin binds to the insulin-receptor on the cell membrane, it causes the membrane to change shape and allows glucose to enter the cell. The cell then uses that glucose for energy.
Insulin is secreted by the pancreas, a gland behind the stomach. The pancreas controls glucose levels in the body by secreting two hormones: insulin, to allow for the uptake of glucose by cells; and glucagon for the release of glucose from the cells back into the bloodstream.
In order for glucose to exit the cell, a familiar process occurs. Only this time the protein glucagon binds to the glucagon-receptor on the cell membrane allowing for the release of glucose from the cells. Together, the hormones insulin and glucagon mean the pancreas regulates blood glucose levels. If your blood glucose levels are not right, you can become quite sick.
Diabetes
In a healthy person, the processes involved in blood glucose regulation are carried out automatically by the body and you are not aware of it. For someone suffering from diabetes, though, the pancreas doesnt properly regulate glucose levels. Diabetes is actually a group of diseases all characterized by high glucose levels in the blood due to the inability of the pancreas to produce sufficient insulin. In some people the pancreas does not produce enough insulin; in some people it doesnt produce any. Diabetes is a chronic condition for which there is no cure. Individuals control diabetes by regulating their own blood glucose levels. When their blood glucose levels are too high, they take insulin, which allows the glucose currently in the blood to be taken up by the cells, thus reducing blood glucose concentration to a normal level. When their blood glucose levels are too low, it means they have to eat something to provide their body with more glucose. Some patients also use glucagon in combination with insulin to regulate blood sugar. Glucagon can sometimes be taken as an alternative to eating something to release glucose from cells back into the bloodstream.
Thousands of Drops; Thousands of Finger Sticks
In order to regulate their blood glucose levels, people with diabetes must measure their blood glucose at least several times a day. This is done by taking a drop of blood, placing it on a paper strip, and inserting the strip into a machine that can measure the glucose level of the blood drop. The drop of blood is usually obtained by pricking a finger with a small needle. After many years of finger pricks, some diabetics can suffer from severe pain and soreness in their fingers. There is currently no alternative for the management of diabetes.
What if it were possible, though, to measure blood glucose concentration without finger-pricking? Dr. Heather Clark of Draper Laboratory in Cambridge, MA is in the process of developing a method that could do just that. It would be like a miniature tattoo, just several millimeters across, that patients would apply to their skin. This “tattoo” would be read using a special monitor to determine glucose concentrations. The monitor would work in the near-infrared or visible light ranges.
How could this be done? Dr. Clark works in the field of nanotechnology. Nanotechnology is technology that takes place on the nano-scale with extremely small units of measurement. One nanometer is one-millionth of a millimeter and one-billionth of a meter. To give you an idea of just how small a nanometer is, the diameter of one human hair is about 60,000 nanometers! Dr. Clarks work since graduate school has focused on nanosensors: sensors monitoring actions on the order of nanometers.
The “tattoo” is actually composed of millions of nanosensors that look like small beads. Each bead is about 120 nanometers in diameter. You can think of it as a rubber bouncy ball – since each bead is about the consistency of rubber – but much, much smaller. This is a bouncy ball on the nanoscale! The sensor is a polymer that has two layers – the rubber ball that is most of the bead, and a very thin layer surrounding it that makes the entire bead biocompatible, or able to interact with cells.
The inner polymer contains two main components: a color-changing dye that is also fluorescent and a component that binds glucose. When no glucose is present, the glucose-binding component interacts with the dye and the bead is yellow and fluorescent. When glucose is present, it is extracted into the sphere and interacts with the glucose-binding component. The glucose-binding component, then, no longer interacts with the dye and the bead turns purple and loses its fluorescence.
With thousands and thousands of similar beads in the same tattoo, it is possible to determine glucose concentration based on the intensity of fluorescence. The greater the intensity, the less glucose is present. The monitor that reads the sensor is like the optical mouse on your computer, with a camera inside. When the monitor is run over the tattoo on your skin, it reads an intensity of fluorescence. A specific intensity matches a specific glucose level.
Dr. Clark envisions the tattoo being placed in the second layer of skin, where it will wear off after about a week as dead skin cells fall off. In fact, it would not be measuring glucose levels in the blood, but glucose levels in the skin. Dr. Clark and her team of researchers have early data that suggests that the two concentrations are comparable, although much work needs to be done is this area.
How It Started
Dr. Clark began her work with nanosensors using the technology to measure sodium flux within a single cell without harming the cell. Several years ago she was asked to make a nanosensor for glucose, and this exciting technology is where her research has taken her so far. The sodium nanosensor is basically a standard sensor used to measure sodium concentrations in blood in a clinical analyzer machine you would find at the hospital brought down to the nano scale. For the glucose nanosensor, Dr. Clark changed the components but used the same basic polymer technology as the sodium sensor.
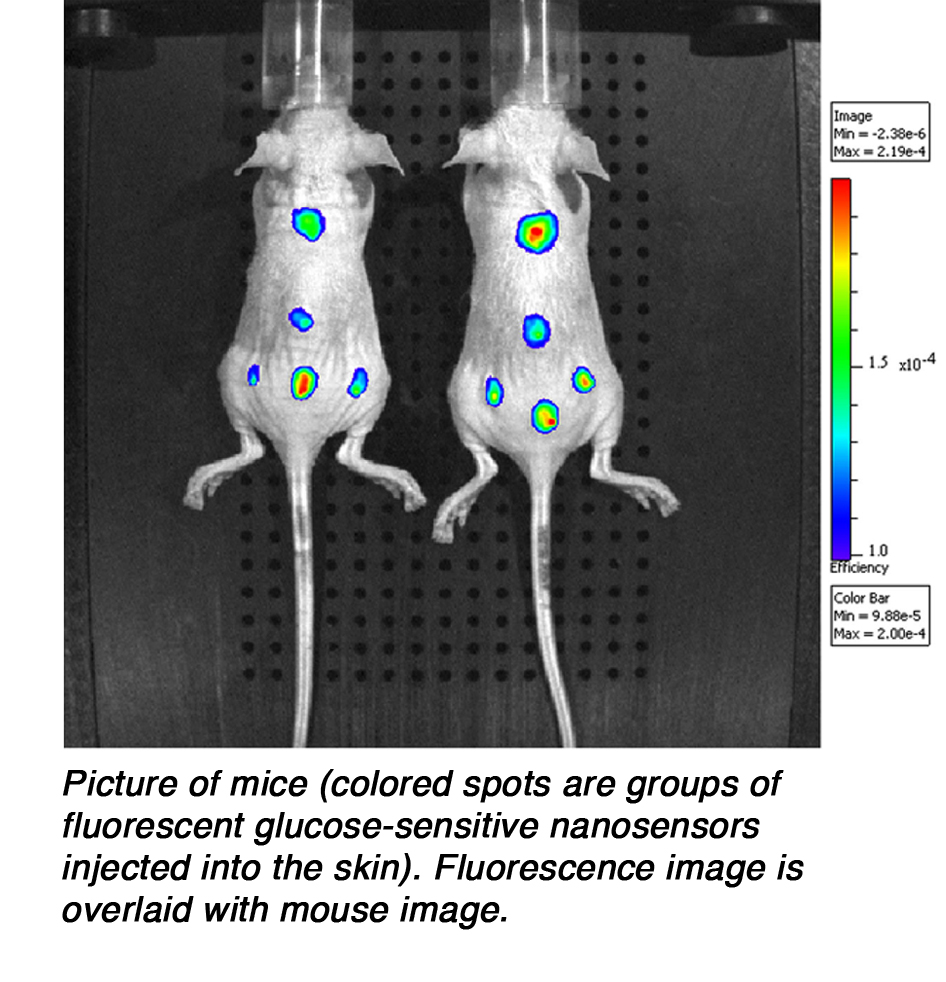 While this is an extremely exciting technology, said Dr. Clark, we are still a long way away from the time when diabetics can apply their own glucose-monitoring tattoo. So far, Dr. Clarks experiments have focused on mice. Mice are first deprived of glucose overnight, and then implanted with nanosensors. The researchers use an animal imager, which is a machine similar to a microscope but made specifically for mice, to watch the intensity fluctuations of the nanosensor, and thus the glucose flux, in the mouse. The traditional finger-prick measurement is also taken and Dr. Clark has shown these two measurements are comparable. So far the results have been very successful. The next step, said Dr. Clark, is to use a mouse model of diabetes. The nanosensors will be inserted into the diabetic mouse, making it possible to watch glucose fluctuations in the mouse over time. When the mouse is given insulin, it would then
be possible to watch the blood glucose levels fall. While this is an extremely exciting technology, said Dr. Clark, we are still a long way away from the time when diabetics can apply their own glucose-monitoring tattoo. So far, Dr. Clarks experiments have focused on mice. Mice are first deprived of glucose overnight, and then implanted with nanosensors. The researchers use an animal imager, which is a machine similar to a microscope but made specifically for mice, to watch the intensity fluctuations of the nanosensor, and thus the glucose flux, in the mouse. The traditional finger-prick measurement is also taken and Dr. Clark has shown these two measurements are comparable. So far the results have been very successful. The next step, said Dr. Clark, is to use a mouse model of diabetes. The nanosensors will be inserted into the diabetic mouse, making it possible to watch glucose fluctuations in the mouse over time. When the mouse is given insulin, it would then
be possible to watch the blood glucose levels fall.
Further studies are also needed to ensure the safety of the technology for human use. There have been no instances of immune reactions in mice, but tests are needed to make sure that the tattoo will not interfere with the immune or other human systems. Research in the lab is ongoing to develop the best polymers and ideal concentrations for the beads.
Jocks, Ions and Tattoos
This technology can also be applied to other molecules and to ions can be used to measure a range of different concentrations. An application currently being explored in Dr. Clarks laboratory is the use of a similar tattoo to monitor sodium concentrations in athletes. Sodium is an important ion in regulating the ratio of fluid to other ions in cells. A high ratio of sodium to fluid in cells indicates dehydration; a low ratio indicates overhydration. It is extremely important for athletes, such as marathon runners, to maintain the right balance of ions like sodium and fluid in their bodies. Dehydration or overhydration can affect their performance and health in dramatic ways. If athletes wore a tattoo that could monitor their cellular sodium levels, it could help them better regulate hydration, preventing both dehydration and overhydration.
To do this, the bead size is changed and the ion-specific polymer core is adapted to detect a different compound.
These are the two technologies we are focusing on right now, said Dr. Clark. But it could be applied to almost any molecule on any scale. With this technology it could be possible to take a closer look at cellular interactions with ions and molecules to determine how each compound interacts with and affects the functioning of cells.
How I Spent My Summer
This summer, Dr. Clark and her laboratory had lots of help from college and high school students interested in biology, chemistry, and engineering and their applications to sensors and nanotechnology. All interns spend their first week exploring sensors and fluorescence before the students are given different projects. The projects are tailored to the interests of the individual students, focusing more on biology for the biologists or chemistry for the chemists, and funnel into the greater project goals. The two high school interns who worked with Dr. Clark this past summer at Draper Laboratory were testing a sensor based on the glucose sensor, but made with much more biocompatible materials. Several college students worked on designing the best way to insert the nanosensor tattoo into the skin of mice.
Dr. Heather Clark is an Independent Investigator at Draper Laboratory in Cambridge, MA. Her current work is focused on developing a nanosensor capable of monitoring glucose levels that could eventually lead to a glucose-monitoring tattoo for diabetics. Dr. Clark has been working with nanosensors since she was a graduate student at the University of Michigan. All through middle and high school, Dr. Clark explains, my passion was art. Then, in high school, I took my first chemistry class, she said, and I was fascinated. She decided to pursue chemistry because she found that she could incorporate the creativity she loved about art into her research. What I love about my job is that I have the ability to be creative in designing and executing experiments. Dr. Clark encourages students to see research both as a science and an art, an outlet to be creative and learn new things at the same time. Dr. Clark has two young children whom she loves to spend time with, when she is not in the laboratory. We love being outdoors: biking, hiking, running, swimming, and skiing.
For more information:
- Dubach, JM., DI Harjes, and HA Clark. “Fluorescent Ion-Selective Nanosensors for Intracellular Analysis with Improved Lifetime and Size.” 2007. Nanoletters, 7: 1827-1831.
- Dubach, JM., DI Harjes, and HA Clark. “Ion-Selective Nano-optodes Incorporating Quantum Dots.” 2007. Journal of the American Chemical Society, 129: 8418-8419.
- Johnson, RD. and LG Bachas. “Ionophore-based ion-selective potentiometric and optical sensors.” 2003. Annals of Bioanalytical Chemistry, 376: 328-341.
To learn more:
About Diabetes:
- American Diabetes Association. http://www.diabetes.org/about-diabetes.jsp
- Centers for Disease Control and Prevention. http://www.cdc.gov/diabetes/
Written by Rebecca Kranz with Andrea
Gwosdow, Ph.D. Gwosdow
Associates
HOME | ABOUT | ARCHIVES | TEACHERS | LINKS | CONTACT
All content on this site is © Massachusetts
Society for Medical Research or others. Please read our copyright
statement — it is important. |
|
|

Dr. Heather Clark

Dr. Clarks team at Draper Laboratory
(l to r): Kristi Shrestha, Matt Dubach, Katie Balaconis, Joe Charest, Liz Balaconis, Michela Meister, Emily Davis, Caroline Maguire, Heather Clark, Emily Conley
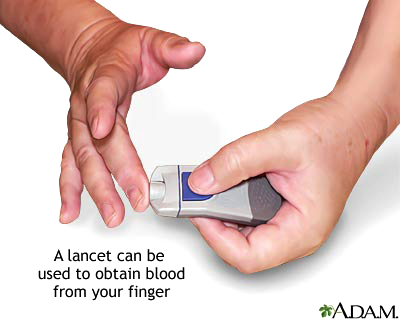
This is a series of five illustrations that shows the steps a person with diabetes goes through to get a blood glucose reading.

Watch the podcast or read the transcript about the glucose monitoring tattoo.
Listen to this radio clip about the glucose monitoring tattoo.
Sign Up for our Monthly Announcement!
...or  subscribe to all of our stories! subscribe to all of our stories!

What A Year! is a project of the Massachusetts
Society for Medical Research.
|
|

